A Danish researcher Thomas Kruse was tasked with solving a bodily problem; treating diabetes and obesity. He was an expert in organic chemistry. Kruse set out on a mission to create GLP 1 with the chemist Jesper Lau and the technician Paw Bloch. They trialed 216 compounds and achieved success with the 217th one! Attaching the fatty acid to the molecule meant that it could now bind to a blood protein called albumin. This protein acted as a shield making the drug more potent at regulating blood sugar and appetite. He first worked on the field of growth hormone secretagogues and PTPase inhibitors before moving to GLP 1!
At Novo Nordisk, the idea was to build on reversible binding to albumin as a solution for the systemic protraction of GLP-1 analogs. The main challenge identified in earlier studies was that strong binding to albumin had a negative impact on the potency of compounds for the GLP-1R, due to competition between binding to albumin and binding to the receptor (42, 46). The theory was that only the free fraction in the plasma that was not bound to albumin would be available to activate the GLP-1R. Successful clinical trials with exenatide and liraglutide led to an increased interest in GLP-1-based therapies. As daily injections are a barrier for some patients with T2D, there was focus on improving convenience, ideally with an effective GLP-1 analog that could be administered once weekly.
He was a medicinal research scientist for 10 years before moving onto peptide chemistry. Semaglutide is a modified form of GLP-1(7–37), where alanine at position 8 is replaced by Aib, lysine at position 34 is replaced by Arg, and a C18 fatty acid chain (Linker) is attached to Lysine at position 26. “I sometimes describe myself as one of Mads Krogsgaard’s guinea pigs,” Thomas jokes. The transition to peptide engineering wasn’t easy, but this reluctant shift would become the foundation for the creation of semaglutide, a medicine now changing millions of lives worldwide. In participants with overweight or obesity, 2.4 mg of semaglutide once weekly plus lifestyle intervention was associated with sustained, clinically relevant reduction in body weight. (Funded by Novo Nordisk; STEP 1 ClinicalTrials.gov number, NCT03548935).
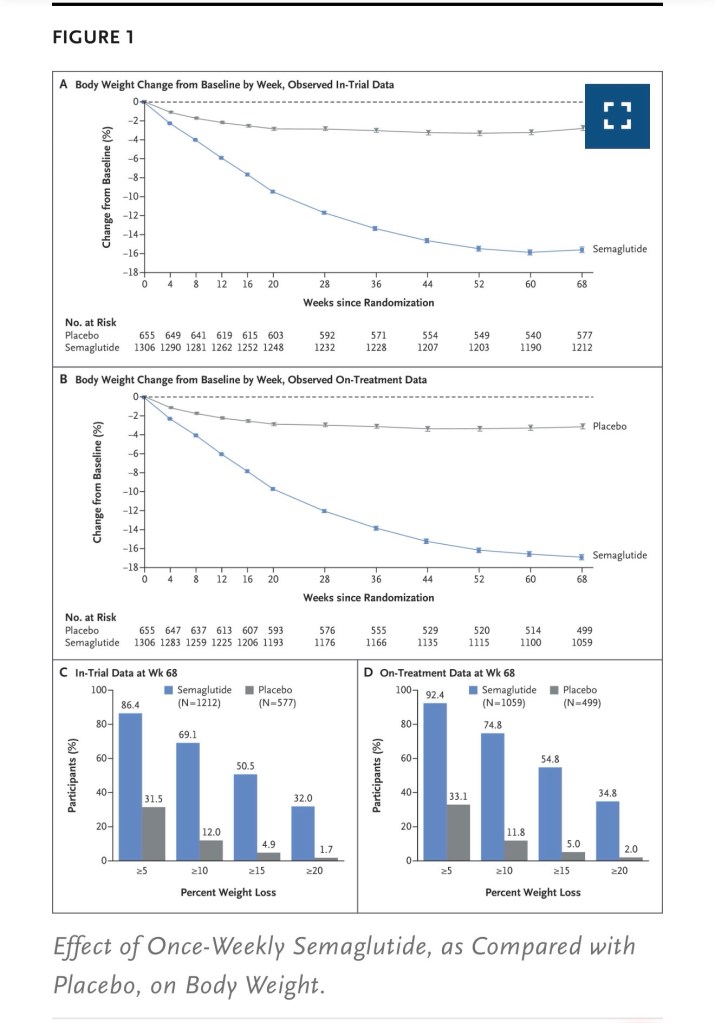
Participants who received semaglutide were more likely to lose 5% or more, 10% or more, 15% or more, and 20% or more of baseline body weight at week 68 than those who received placebo (P<0.001 for the 5%, 10%, and 15% thresholds; the 20% threshold was not part of the statistical testing hierarchy). The extended benefits of Semaglutide are impressive to say the least; it has an extended exposure-OW (stands for once weekly) sc or once daily oral regimen, hypoglycaemia is minimised- there is stabilised receptor activity, less injecting-patient compliance increases, manufacturing wise- there is a high product purity on a commercial scale.
Question Is treatment with semaglutide associated with weight loss outcomes similar to those seen in results of randomised clinical trials?
Findings In this cohort study of 175 patients with overweight or obesity, the total body weight loss percentages achieved were 5.9% at 3 months and 10.9% at 6 months.
Meaning Semaglutide treatment in a regular clinical setting was associated with weight loss similar to that seen in randomised clinical trials, which suggests its applicability for treating patients with overweight or obesity.
Comparatively Studying and Distinguishing Semaglutide to Other Drugs
- In their meta-analysis of 16 studies totaling 5997 patients, Karimi et al. [56] compared the effectiveness of semaglutide versus liraglutide, dulaglutide, or tirzepatide.
- The data indicated that semaglutide reduced HbA1c values compared to liraglutide. However, no significant differences were observed regarding fasting glucose, BMI, and weight change between semaglutide and liraglutide.
- Semaglutide was more effective than dulaglutide in reducing HbA1c levels and fasting glycemia although no significant differences were found in body weight and BMI changes.
- When compared to semaglutide tirzepatide was more effective at reducing HbA1c levels without any clear superiority in changes in body weight and fasting blood glucose.
- Regarding drug transition, moving from liraglutide to semaglutide did not significantly impact HbA1c levels but did induce weight loss and reduce fasting blood glucose.
- Conversely, transitioning from dulaglutide to semaglutide did not modify HbA1c and weight changes.
The study concludes that analyses consistently demonstrate superior efficacy of semaglutide compared to liraglutide in reducing both HbA1c levels and weight. Moreover, it is more effective than dulaglutide in diminishing fasting glycemia. On the other hand, tirzepatide reduces HbA1c values more effectively than semaglutide.
Question What factors are associated with semaglutide initiation in individuals with obesity who do not have diabetes?
In this cohort study, our analysis of a large commercial health insurance claims database identified several key sociodemographic, health care, and clinical factors that were associated with semaglutide initiation in individuals with obesity and without diabetes. Certain medication classes, age, sex, industry types, and plan structures were associated with semaglutide uptake. The subgroup of individuals with obesity but without diabetes has been often omitted from discussions around equity in obesity treatment. Individuals that have metabolically healthy obesity still exhibit an elevated risk of developing cardiometabolic diseases.25 Older paradigms of obesity treatment conceptualized obesity as a risk factor for further metabolic disease, and while obesity does increase risk, it is now considered to be an independent disease. However, clinicians greatly underprescribe antiobesity medications compared with antidiabetic medications.26 Additionally, individuals with higher reported BMI at baseline were more likely to initiate semaglutide, a finding that reflects the disconnect between clinician behavior and treatment guidelines. While antiobesity medications are recommended for individuals with a BMI of 30 or greater or a BMI of 27 with a weight-related comorbidity,27 many clinicians opt to restrict treatment to lifestyle and behavioral therapy if an individual has a BMI less than 35. This trend persists, despite the growing body of evidence concluding that individuals with class I obesity often develop class II and III obesity over the life course.28
- Use of other medications was found to be a significant factor associated with semaglutide uptake. Antidepressant use is common in individuals with obesity because there is a bidirectional biological association of depression with obesity.29
- Supply shortages have also presented a major barrier to access for individuals prescribed a GLP-1 receptor agonist.32
- Individuals covered by exclusive provider organization and HMO plan types were less likely to initiate semaglutide therapy. HMO plans require patients to select a primary care physician, which may result in increased utilization of care. Patient financial responsibility depends on the actuarial value and benefit design of the insurance plan.37
- Employer industry type was associated with access to semaglutide as well, which may be due to the relative earnings in different lines of work. According to the Bureau of Labor Statistics, as of April 2024 individuals employed in the financial and professional services industries earned $45.30 and $41.82 per hour, while those in the retail industry earned $24.25 per hour.43 Both semaglutide and tirzepatide have high list prices, hovering around $1000 per month.44 Manufacturer coupons can reduce the cost burden by $225 to $500 per month for patients paying out of pocket.45 Prices appear to be elevated for those accessing the drugs for nondiabetes indications, with the estimated monthly price net of discounts for semaglutide for diabetes being $344 to $411 lower than that of semaglutide for weight management.46,47 For those on public payer plans, which represents those particularly at-risk of mortality and morbidity from obesity,48 coverage is highly variable. Historically, Medicare Part D plans were forbidden from covering pharmacotherapies for weight management indications, while being permitted to cover those same active ingredients to treat diabetes. While policy changes have been proposed to expand access,49 there is still pressure to exclude these medications for budgetary concerns.
The findings may help clinicians better understand which patients are less likely to access obesity treatment, enabling more equitable care. For policymakers, the results underscore the need to address structural barriers, such as expanding insurance coverage of weight loss medications to promote access to effective obesity treatments. Overall, this study highlights the need to investigate antiobesity medication use in varied populations as the obesity treatment landscape changes with the introduction of novel treatments. One limitation of this study is its reliance on claims data, which lags behind prescribing and health care provision by 3 to 6 months while claims are adjudicated. Therefore, the most recent data may have been missing prescriptions.
Semaglutide Treatment in the Long-Term Run
In patients treated with semaglutide, weight loss continued over 65 weeks and was sustained for up to 4 years. Clinically meaningful weight loss occurred in both sexes and all races, body sizes and regions. Semaglutide was associated with fewer serious adverse events. For each BMI category (<30, 30 to <35, 35 to <40 and ≥40 kg m−2) there were lower rates (events per 100 years of observation) of serious adverse events with semaglutide (43.23, 43.54, 51.07 and 47.06 for semaglutide and 50.48, 49.66, 52.73 and 60.85 for placebo). Semaglutide was associated with increased rates of trial product discontinuation. Discontinuations increased as BMI class decreased. In SELECT, at 208 weeks, semaglutide produced clinically significant weight loss and improvements in anthropometric measurements versus placebo. Weight loss was sustained over 4 years.
SELECT-Semaglutide Effects on Cardiovascular Outcomes in People with Overweight or Obesity trial
Semaglutide and Eating Disorders
This study aimed to identify the prevalence of prescription weight loss medication use among boys and men, describe the sociodemographic differences between those who did and did not report use, and explore differences in eating disorder attitudes and behaviors between those who did and did not report use. Data from 1543 boys and men from Canada and the United States aged 15 to 35 were analyzed. The prevalence of prescription weight loss medication use in the past 12 months was estimated. Fisher’s exact tests and independent samples t-tests were used to determine the differences in sociodemographic identifiers and eating disorder attitudes and behaviors between those who did and did not use prescription weight loss medication. Among the sample, 1.2 % (n = 19) reported use of prescription weight loss medication in the past 12 months. Those who reported use of prescription weight loss medication were significantly older and had significantly higher body mass index compared to those who did not report use.
Any loss of control while eating, binge eating, and purging via vomiting in the past 28 days were all more common among those who reported the use of prescription weight loss medication. Eating disorder psychopathology was also significantly higher among those who reported the use of prescription weight loss medication. These preliminary findings underscore that eating disorder attitudes and behaviors may be more prevalent among boys and men who use prescription weight loss medication, emphasizing the need for more research to understand these novel findings.
Sources and Information:
https://www.novonordisk.com/disease-areas/obesity/the-story-of-semaglutide.html
https://annualreport.novonordisk.com/2025/introducing-novo-nordisk/semaglutide.html
https://www.nejm.org/doi/full/10.1056/NEJMoa2032183
https://jamanetwork.com/journals/jamanetworkopen/fullarticle/2796491
https://pmc.ncbi.nlm.nih.gov/articles/PMC6474072/
https://jamanetwork.com/journals/jamanetworkopen/fullarticle/2829420
https://www.nature.com/articles/s41591-024-02996-7
https://www.sciencedirect.com/science/article/pii/S1471015325000789
Image credit
https://unsplash.com/photos/medicine-box-and-injection-pens-for-medical-use-aLCcungJbrc